As a first step to making your own products, we highly recommend enrolling on our free online course (over 17,500 students) www.learncosmeticformulation.com which comes with a PDF certificate of completion. It’s online so you can start anytime and work at your own pace, after each Unit is released.
Why do we need a preservative?
A preservative is essential to help prevent microbes (bacteria, mold, and yeast) growing. Preservatives stop growth by acting on spores when they germinate and killing cells (usually by disrupting cell membranes) or by making the product hostile to growth.
In 1947, in New Zealand, unsterilised talc caused 25 cases of Tetanus, and 4 fatalities.
When do we need a preservative? Do scrubs, liquid soap and anhydrous products need preserving?
If your product contains water (including hydrosols, floral water, aloe vera, goat’s milk which all contain water) or will come into contact with water (eg a scrub used with wet fingers) a broad spectrum preservative is needed.
Cold process soap (made with sodium hydroxide) does not need a preservative. For other high pH products such as liquid soap, generally if the pH is above 10 a preservative may not be required. If the pH is below 10, liquid germall plus can be used (despite the supplier recommended use below pH8). Alternatively, Suttocide A (see below for downsides) or Glydant Plus can be used.
For anhydrous (no water) products or scrubs, if water may be introduced to the product or the product used in a humid bathroom then a preservative is advisable. An expert microbiologist advises that if trying to preserve an anhydrous product (including all oil+sugar/salt scrub) the oil soluble preservative will get locked in the oils so will not reach any water, if water was introduced into the product. So if you added an oil soluble preservative then that preservative will stay in the oils and not move over to where the water is located to protect that water against bacteria and mould so would be useless. So contrary to what you may have read, we should really use a water soluble preservative in an anhydrous product which means we need to add an emulsifier to get that preservative mixed in properly with the oils.
If you choose not to include a preservative your product may last up to 5 days if stored in the fridge.
Can we use vitamin E, rosemary extract or grapefruit seed extract to preserve?
It’s a common myth that anti-oxidants such as vitamin E, grapefruit seed extract and rosemary extract are preservatives but they are actually anti-oxidants. Oxidation of oils and butters leads to rancidity and anti-oxidants slow down this process. These anti-oxidants do not prevent bacteria, yeast, or mold from spoiling your product.
How to tell if your product is sufficiently preserved?
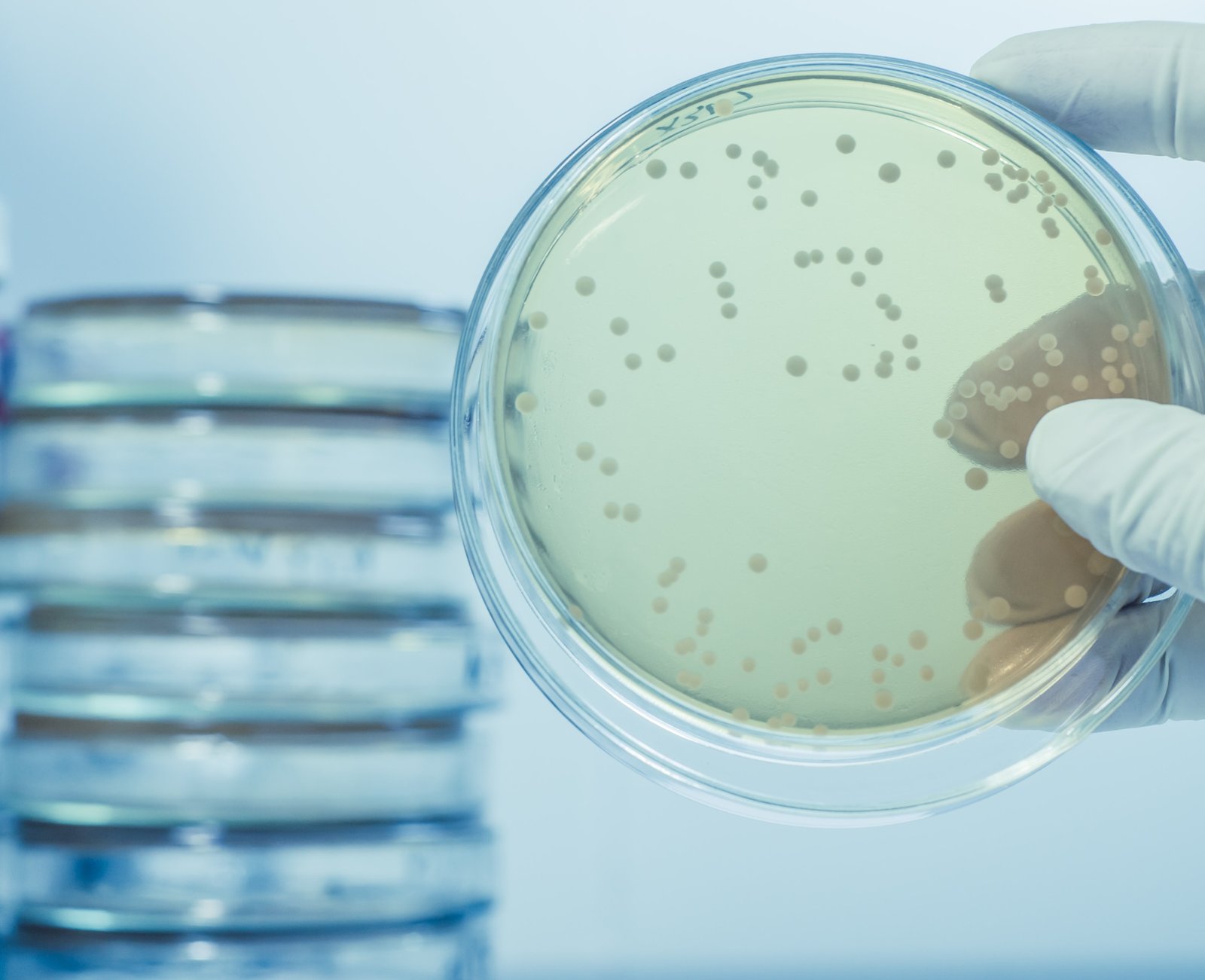
We cannot tell by sight or smell if our product is preserved properly. For example, if 100,00 bacteria is introduced into less than one quarter of a teaspoon of water, the water will smell and look the same as sterile water. Most cosmetics tested have counts ranging into the tens of thousands or millions of cells per milliliter have subtle or no aesthetic differences from sterile samples. As home test kits are very inaccurate the only way to assess if the preservative is working is by a challenge test or preservative efficacy test. In this test, microorganisms are introduced (1 x 10 to the power of 6 bugs/gram) into the product on day 1 with a re-challenge at day 21. This is performed on a mixed culture of organisms:– staphylococcus aureus – gram positive
– escherichia coli – gram negative
– pseudomonas aeruginosa – gram negative
– candida albicans – yeast
– aspergillus niger – mould
The total test period is 28 days with plate counts being run on 0, 1, 2, 7, 21 and 28 days. The preservative is judged to have passed if it has the ability to kill the introduced bugs and at the 21 days rechallenge retains its activity. An unpreserved control sample must be run and must fail this test.
Factors which greatly impact the effectiveness of your chosen preservative system
It is not sufficient to just add a preservative blend and trust that this is enough. The following factors need to be considered:-
- pH. The finished pH of the product will have a major impact on the efficacy of the preservative. Bacteria prefer pH 5.5 to 8.5, the typical pH range for bacterial growth is pH 4 to 9. For yeasts and filamentous fungi growth generally occurs within a range of pH 3 to 10 but pH 4-6 is where there is the most growth.
- Reducing the amount of unbound water in your formulation will help to inhibit microbial growth is used by some formulators as part of the “hurdle approach”. This can be accomplished with the use of ingredients such as pentylene glycol, propanediol.
- Microbes need food to grow so it is best to limit “bug food”. Examples include fruit, botanicals, tea, lecithin, mineral water, milk of any kind, honey, hydrosols, floral waters, aloe vera, extracts, protein, clay, powders, starches etc. It is possible to use, for example, 50% aloe vera or hydrosol in a formula and still pass preservative tests if the formula is carefully formulated with that in mind and the supplier’s copy of the micro testing for the batch of aloe/hydrosols in the formula (could be in the Certificate of Analysis) shows that ingredient is less than 100 cfu/gram or ml and no staphylococcus aureus, candida albicans or gram negative bacteria!
- The preservative should be protecting the water phase so your preservative system will be affected negatively if there are solid particles for the preservative to adsorb onto, for example, iron oxides, mica, zinc oxide, titanium dioxide.
- Check below whether the combination of preservatives you choose will make a broad spectrum blend. If your preservative is not listed on this page after joining our discussion group you can look up the preservative in this post. do check for it here.
- Use good GMP to include microbiological testing of raw ingredients and process water, equipment sanitization.
- Add 0.2% disodium EDTA, a chelator, into the heated water phase. Other chelators are much weaker in comparison
- Switch to packaging which the customer cannot contaminate easily – jars are the worst for contamination.
- Use distilled, deionised or purified water, not tap/faucet or mineral water
- If your water has not been micro checked, heat and hold your water phase at 75°c/167°f for 20 minutes. This will kill some of the non-endospore forming bacteria. (If your preservative can withstand heat put it in the heated water phase rather than the heated oil phase. This improves preservative contact with the water phase so that it is not partitioned in the water-oil interface). To access a discussion between expert chemists and microbiologists on why the heat and hold is needed, after joining our group click here.
Preservative free – glycerin, alcohol and honey as preservatives – the hurdle approach?
Many companies are advertising their products as “preservative free”. In most cases their products do contain preservatives but as they also have a dual function companies are relying on the ingredient’s other function when they are making their claim. Further, some companies are using the “hurdle approach” to help preservative their product. Examples below:-
- As mentioned above, a very high or very low pH can make the product more hostile for some microbes.
- 70% glycerin is an effective preservative however it is very sticky at that concentration. Other glycols, such as propanediol, caprylyl glycol, pentylene glycol may also be used but can affect viscosity, stability and affect skin feel.
- Ethanol (not vodka, instead use 190 Everclear alcohol or skin safe cosmetic use denatured alcohol) anything containing 20-25% ethanol is self preserving. Alcohol is astringent so may not be suitable for those with sensitive nor dry skin.
- Honey once diluted becomes a source of microbial contamination.
- Grapefruit seed extract (not recommended) is chemically derived from the seeds of citrus fruits. It is made by reacting with ammonia, so is more like a quat in some ways. There are concerns that the limited preservative properties grapefruit seed extract may have are in fact due to added preservatives like parabens.
Which preservatives to use?
As a general rule it is always a good idea to mix up your preservatives, as they all have their strengths and weaknesses in what they kill, so a combination can give you broad spectrum protection, and keep the overall levels of each down low, which helps with both stability and irritancy. Most of the preservatives listed below are a mixture of preservatives.
Use a preservative blend which is broad spectrum. This means it guards against (a) gram positive bacteria, (b) gram negative bacteria; and (b) fungi. Usually preservative blends only guard against one or two of these so are not broad spectrum.
If you have decided to make “all-natural” skin care products – you will not find a product on the market that is all-natural, safe for use and will protect your product from the wide range of microbes and fungi out there. Essential oils such as tea tree have been suggested, however, the percentage required to be effective as a preservative would not be safe to use on the skin. With the more newer more “natural” type preservatives, yeast and mold are frequent issues.
To make matters worse, ingredient suppliers often advertise their “natural” preservative as broad spectrum when it is far from the case. Click here for a list of worldwide cosmetic ingredient suppliers.
Below is a list of commonly used preservatives. As there is no agreed definition of “natural” and “organic”, and it takes a great deal of technical knowledge and time to investigate each ingredient fully, most formulators look to organisations (USDA Organic, Wholefoods, NPA, COSMOS etc.) who have analysed and rated ingredients along several parameters, including the origin, sustainability and processing. It is advisable to look up the preservative below on the organisation’s website to see if the ingredient meets their particular standard.

Trade name(s) of preservative: Benzostat
INCI name: Caprylhydroxamic Acid (and) Benzyl Alcohol (and) Glycerin
Usage and phase: 1% water phase. (Depending on the temperature it is stored at, if the benzostat is not liquid, it will need melting at around 45°C with mixing before adding to the formula).
Works within pH range: Supplier states it has a wide pH range
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Yes
– fungi? Yes
Further details: The supplier provided some of the information below. Unfortunately this preservative is not sold by DIY suppliers. Benzostat can interact with iron found in clay, bentonite, silicates, etc producing a orange colour.
Trade name(s) of preservative: Euxyl K940
INCI name: Phenoxyethanol, Benzyl Alcohol, Ethylhexylglycerin, Tocopherol
Usage and phase: 1% Water phase
Works within pH range: up to 12
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Yes
– fungi? Yes
Further details: sold by Plena Natura
Trade name(s) of preservative: Liquid germall plus
INCI name: Propylene Glycol (and) Diazolidinyl Urea (and) Iodopropynyl Butylcarbamate
Usage and phase: 0.5% Cool down
Works within pH range: 3-8 but can be used at higher pH, for example, pH 10
Natural or synthetic? Synthetic
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Yes
– fungi? Yes
Further details: This effective and easy to use preservative is a favourite amongst DIYers including swiftcraftymonkey. In the US, do not use in aerosols/sprays and in the EU do not use in body creams/lotions.
Trade name(s) of preservative: Phenoxyethanol
INCI name: Phenoxyethanol
Usage and phase: 0.6-1% Cool down
Works within pH range: 3-10
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Somewhat
– gram negative bacteria? Yes
– fungi? No
Further details: Phenoxyethanol is stable up to 85°C (185°F)and is soluble in most oils. It is also soluble in water from 0.5 to 2.67 grams per 100 grams of water. It is miscible with propylene glycol and glycerin. Phenoxyethanol is inactivated by highly ethoxylated compounds including polysorbates so do not use with surfactants. In surfactant solution systems, the water must be saturated with phenoxyethanol for activity. If the level is too low, it acts as a nutrient for bacteria. However, it can cause viscosity issues in detergent products, Phenoxyethanol is slightly volatile so it will protect the “head space” or vacant air space above the product in the bottle.
Trade name(s) of preservative: Geogard 221, Cosgard
INCI name: dehydroacetic acid and benzyl alcohol
Usage and phase: 1% Any
Works within pH range: 2-6
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Poor
– fungi? Somehwat
Further details: Not approved for aerosol use. Over time benzyl alcohol oxidises to benzaldehdye resulting in an almond odour. The dehydroacetic acid protects against fungi and can sometimes cause a yellowing effect which can be limited by adding antioxidants e.g. BHT or sodium metabisulfite. When used with anionic surfactants such as ether sulfates and cocamidopropyl amides, there may be a darkening effect.
Trade name(s) of preservative: Optiphen, GFphen PCG. Mikrokill COS, GFguard COS.
INCI name: Phenoxyethanol (and) Caprylyl Glycol. Phenoxyethanol (and) Caprylyl Glycol (and) Chlorphenesin).
Usage and phase: 1% Cool down
Works within pH range: 4-8
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Yes
– fungi? Somewhat
Further details: Can cause emulsion instability. To strength your emulsion: (a) use two reliable and stable emulsifiers such as ceteareth-20 and polysorbate 60 at the right concentration, and (b) add extra cetyl alcohol and 0.3% xanthan gum; and (c) remove any electrolytes; and (d) use high shear (stick blending). As this preservative contains phenoxyethanol the details above regarding that ingredient also apply. For an all water solution as the caprylyl glycol (or ethylhexylglycerin in the case of Euxyl PE9010) is oil soluble, a solubiliser may be required.
Trade name(s) of preservative: Optiphen plus
INCI name: Phenoxyethanol (and) Caprylyl Glycol (and) Sorbic Acid
Usage and phase: 1% Cool down
Works within pH range: 4-6
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Yes
– fungi? Somewhat
Further details: See the above comments for optiphen. The sorbic acid added to make optiphen plus is negligible (6% of the blend) at 0.06% if 1% optiphen plus is in the formula. Therefore the addition of sorbic acid is insignificant so this preservative performs similarly to optiphen.
Trade name(s) of preservative: Preservative 12, Euxyl® PE 9010, Ethox, GFpreserve POG
INCI name: Phenoxyethanol (and) ethylhexylglycerin
Usage and phase: 1% Cool down
Works within pH range: up to 9
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Yes
– fungi? Somewhat
Further details: See the comments for Optiphen above.
Trade name(s) of preservative: Naticide
INCI name: fragrance/parfum
Usage and phase: 1% Cool down
Works within pH range: below pH 5
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Poor
– gram negative bacteria? Poor
– fungi? No
Further details: Very expensive and has a vanilla/almond scent. Many users report irritation.
Trade name(s) of preservative: Microgard, Geogard Ultra, Neodefend
INCI name: gluconolactone (and) sodium benzoate
Usage and phase: 2% Cool down (dissolve in water before adding)
Works within pH range: below pH 5
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? No
– fungi? Partially
Further details: Frequently causes pH drift downwards. Cover the gram negative bacteria gap with phenoxyethanol or 1% phenyl ethyl alcohol
Trade name(s) of preservative: Mikrokill ect, preservative eco, geogard ect, plantaserv M
INCI name: Benzyl Alcohol & Salicylic Acid & Glycerine & Sorbic Acid
Usage and phase: 1.1% Cool down
Works within pH range: below 5.5
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Somewhat
– fungi? Poor
Further details: Over time benzyl alcohol oxidises to benzaldehdye resulting in an almond odour
Trade name(s) of preservative: Dermosoft® 1388 ECO
INCI name: Glycerin, Aqua, Sodium Levulinate, Sodium Anisate
Usage and phase: 3.5% Water
Works within pH range: 5-5.3 (this preservative crystallises out as it becomes insoluble and can cause emulsion separation if the pH goes below 5. However, the preservative should be below 5.5 to be active)
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Moderate
– gram negative bacteria? Moderate
– fungi? No
Further details: needs careful formulating to be within the recommended pH range. Monitor the pH over time.
Trade name(s) of preservative: Suttocide A
INCI name: Sodium Hydroxymethylglycinate
Usage and phase: 0.3% Cool down phase before fragrance oil or essential oils or salt are added
Works within pH range: 3.5-12
Natural or synthetic? Synthetic
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Yes
– fungi? Moderate
Further details: One of the few preservatives which can handle a high pH. The preservative itself also has a high pH. Citral (a component of some essential and fragrance oils e.g. citrus) will react with Suttocide A changing the colour over time to a light red.
Trade name(s) of preservative: Dermosoft GMCY
INCI name: Glyceryl Caprylate
Usage and phase: up to 1% Water
Works within pH range: 4-7
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? No
– fungi? No
Further details: Unsuitable for use with surfactants. Can cause emulsion instability so it is advisable to follow the tips above under Optiphen in order to strengthen your emulsion. Oil soluble so needs a solubiliser in aqueous products.
Trade name(s) of preservative: Various
INCI name: Benzoic acid or potassium benzoate or sodium benzoate
Usage and phase: 0.3% Cool down
Works within pH range: below 5
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Poor
– fungi? Yes
Further details: Can cause irritation. More stable than potassium sorbate and less pH sensitive. Incompatible with quaternary compounds and non ionic surfactants.
Trade name(s) of preservative: Various
INCI name: Sorbic acid or potassium sorbate
Usage and phase: 0.3% Cool down
Works within pH range: below 5
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Somewhat
– gram negative bacteria? Somewhat
– fungi? Yes
Further details: Can cause irritation. Sensitive to oxidation resulting in discoloration and unstable at temperatures above 38°C. Some testers notice a petroleum odour and experience flushing and reddening of face when sorbates are used in leave on products probably due to peripheral vasodilation effect of sorbates. Sorbic acid is sensitive to oxidation resulting in discoloration and a potential petroleum odor.
Trade name(s) of preservative: Phenonip
INCI name: Phenoxyethanol (and) Methylparaben (and) Ethylparaben (and) Butylparaben (and) Propylparaben
Usage and phase: 1% heated water phase (dissolves around 60˚C to 70˚C)
Works within pH range: 3.5-7.5
Natural or synthetic? Synthetic
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Yes
– fungi? Yes
Further details: Popular effective preservative. Can be inactivated by some non-ionic emulsifiers, such as polysorbates, ceteareth-20 and strong hydrogen bonders such as ethoxylated compounds, cellulose gums, lecithin. Parabens are very oil soluble so tend to migrate to the oil phase, where their activity is greatly diminished, particularly if there are a lot of esters and vegetable oils so it may not be suitable for high oil loads in an emulsion. Parabens must be added to the water phase or they will remain in the oil phase and have reduced activity against bacteria and fungi.
Trade name(s) of preservative: Leucidal Liquid SF, Leucidal Liquid and Leucidal Complete
INCI name: Lactobacillus Ferment. Leuconostoc/Radish Root Ferment Filtrate. Leuconostoc/Radish Root Ferment Filtrate, & Lactobacillus & Cocos Nucifera (Coconut) Fruit Extract
Trade name(s) of preservative: PhytoCide Aspen Bark Extract Powder
INCI name: Populus tremuloides Bark Extract
Trade name(s) of preservative: Amticide Coconut
INCI name: Lactobacillus & Cocos Nucifera (Coconut) Fruit Extract
Trade name(s) of preservative: Phytocide elderberry OS
INCI name: sambucus nigra fruit extract
Natural or synthetic? Natural
Protects against:
– gram positive bacteria? Poor
– gram negative bacteria? Poor
– fungi? No
Further details: These preservatives are from the same supplier and are listed together for ease of reference.
Trade name(s) of preservative: Germaben II
INCI name: Propylene Glycol (and) Diazolidinyl Urea (and) Methylparaben (and) Propylparaben
Usage and phase: 1% Cool down
Works within pH range: 3-7.5
Natural or synthetic? Synthetic
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Yes
– fungi? Yes
Further details: For emulsions with an oil phase of about 25% or less. Popular effective preservative. Can be inactivated by some non-ionic emulsifiers, such as polysorbates, ceteareth-20 and strong hydrogen bonders such as ethoxylated compounds, cellulose gums, lecithin.
Trade name(s) of preservative: Germaben II-E
INCI name: Propylene Glycol (and) Diazolidinyl Urea (and) Methylparaben (and) Propylparaben
Usage and phase: 1% Cool down
Works within pH range: 3-7.5
Natural or synthetic? Synthetic
Protects against:
– gram positive bacteria? Yes
– gram negative bacteria? Yes
– fungi? Yes
Further details: Use Germaben II-E (as opposed to Germaben II) when making emulsions that have 25% oil or more. Popular effective preservative. Can be inactivated by some non-ionic emulsifiers, such as polysorbates, ceteareth-20 and strong hydrogen bonders such as ethoxylated compounds, cellulose gums, lecithin.